Key takeaways
- A Certificate of Analysis (CoA) from a qualified lab confirms a product’s identity, potency, purity, and quality against a stated specification, following established quality standards.
- For supplements, a CoA verifies label claims, screens for contaminants, and links results to a specific lot for traceability and consumer confidence. You may see different types of CoAs depending on whether they cover raw materials, finished goods, or specialized testing.
- To review a CoA, check the lot number, compare the results to the stated ranges, verify the lab and methods, and look for an authorized signature—steps that help you evaluate CoA accuracy at a glance.
- A Certificate of Analysis supports quality at every step, from sourcing ingredients to manufacturing and internal reviews. Because creating CoAs follows controlled procedures, the results are reliable, repeatable, and easy to trace across batches.
- Third-party testing identifies the independent lab that runs the analysis. The CoA is the document that shows those results, and it shouldn’t be confused with a safety data sheet (SDS), which covers handling and storage guidance.
- InfiniWell works with qualified laboratories, follows practices aligned with Good Manufacturing Practice (GMP) standards, and provides access to batch-specific CoAs so you can compare real data and choose confidently.
Plenty of supplements look impressive on the shelf, but looks alone don’t say anything about how well a product was tested. And with news stories calling out mislabeled formulas and weak quality control, relying on the label alone isn’t enough.
That’s where a CoA comes in. It’s the document that shows a batch was actually tested by a qualified lab and that the results met the brand’s stated standards.
If you’ve ever searched “what is a CoA?” or “what is a certificate of analysis,” this guide offers a clear, straightforward explanation so you can read CoAs with confidence.
CoA meaning: What a Certificate of Analysis is
A Certificate of Analysis is a data report created by a qualified laboratory, showing how a batch performed when tested against the brand’s established specification. In short, it's proof of what's in the bottle—and what isn't.
CoAs appear throughout manufacturing and supply chain workflows, especially for raw materials and finished goods. Each document is a snapshot of test data, signed by an authorized analyst, showing identity, strength, purity, and overall quality.
In supplements, that often includes ingredient verification, potency, and checks for things manufacturers work to avoid, such as heavy metals or microbes, to support quality and regulatory compliance.
Different types of CoAs
CoAs can look slightly different depending on where they’re used in the production process. Common examples include:
- Raw material CoAs: Confirm the identity and purity of each ingredient before manufacturing begins.
- In-process CoAs: Document testing performed during production to ensure the batch stays within specification.
- Finished-product CoAs: Show final potency, purity, identity, and microbial results for the completed batch.
- Specialized CoAs: Focus on specific parameters, such as heavy metals, allergens, or microbial limits, when additional verification is needed.
- Third-party CoAs: Issued by an independent lab to add an extra layer of transparency and validation.
Why CoA standards matter
Standards bodies such as the International Organization for Standardization (ISO) outline frameworks for labs and quality systems, and many manufacturers follow GMP guidelines.
Together, these create the quality assurance backbone that makes a CoA meaningful because the data is generated under controlled, validated methods.
Why CoAs matter for supplements
Supplements are about trust. Seeing a label is one thing; knowing the product was actually tested is another. A CoA helps you confirm that a batch went through qualified laboratory testing and that the results met the stated specification.
Here’s why that matters:
-
Identity and potency: A CoA confirms that the ingredients and amounts listed on the label match what’s in the bottle.
-
Purity and safety: It shows the batch was screened for unwanted substances, such as heavy metals or microbes, supporting consumer confidence and overall product safety.
-
Quality culture: It signals that the brand prioritizes quality control, proper documentation, and consistent production standards.
-
Traceability: It links results to a specific lot, test methods, and dates, which supports responsible risk management and internal audits.
While regulation varies, reputable businesses use CoAs as part of their everyday quality practice. That consistency is good for your peace of mind and your wellness routine.
What a CoA looks like
Most CoAs follow a familiar format so you can scan them quickly. You'll typically find:
-
Product and batch details: Product name, lot/batch number, manufacturing date, and sometimes expiration or retest date.
-
Sample information: Where and how the sample was retrieved, plus the specification it was tested against.
-
Test methods: Method names or references and the laboratory that performed the work.
-
Results and units: The numerical data with acceptable ranges. This is the heart of the document.
-
Pass/fail assessment: Whether each test met the specification.
-
Sign-off: An authorized signature and date, often with a certificate number and quality control review stamp.
Depending on the product, you might also see notes about technology used (like HPLC, ICP-MS), storage conditions, and disclaimers.
What does "CoA" stand for in business?
In business, CoA still refers to the same Certificate of Analysis described above, used across many industries. The details tested may change from one sector to another, but the purpose stays the same: a CoA verifies that a product or material was tested and met its specification.
Here’s how different industries rely on CoAs:
-
Ingredient and raw material suppliers: Verify identity, purity, and quality before materials enter manufacturing.
-
Food and beverage companies: Document allergen checks, microbial limits, and ingredient verification to support food safety programs.
-
Cosmetics and personal care: Confirm purity, heavy metal levels, and compliance with applicable quality standards.
-
Specialty chemicals and agriculture: Verify concentration, composition, and safety parameters before distribution.
-
Laboratories and testing services: Issue CoAs to communicate validated results between businesses.
-
Health-related and certain drug manufacturing steps: Use CoAs to document that materials meet defined specifications.
-
Nutritional supplements: Review CoAs for both raw materials and finished batches to support transparency, traceability, and customer confidence.
Across all of these sectors, a CoA functions as a cross-industry quality document that supports regulatory expectations, internal controls, and consistent production standards.
How CoAs are created
A CoA only matters if the data behind it is accurate. That’s why labs follow strict quality standards and regulatory expectations, whether the work is done for supplements, food products, or other industries that rely on verified testing.
Here’s the general process:
-
Sampling: A batch sample is collected according to documented procedures to avoid contamination or human error.
-
Testing: Accredited labs run validated methods to check identity, potency, purity, heavy metals, allergens, and microbiological markers.
-
Review: Analysts confirm the data meets the product’s specification ranges.
-
Documentation: Results are compiled into a CoA, including acceptable ranges, actual values, methods used, and the authorized sign-off.
-
Delivery: The final CoA is released to the manufacturer or posted for customers to review.
CoA formats vary slightly by country or industry, but the core elements (methodology, results, and accuracy) remain consistent. These practices help reduce errors and ensure every batch meets applicable standards.
How to read a CoA
A CoA can look technical at first glance, but most follow the same structure. Once you know the key sections to focus on, it becomes easy to evaluate how a batch performed and whether it met the stated standards.
-
Confirm the batch: Match the lot number on your product to the CoA’s lot number. If it doesn’t match, it isn’t the correct document.
-
Review the specification: Look for the acceptable range for each test (for example, “≥ 95%” or “NMT 0.1 ppm”) and make sure the results fall within that range.
-
Check the methods and lab: A solid CoA lists the methods or references used and identifies the laboratory. ISO-accredited labs and validated methods add confidence.
-
Read the results: Focus on identity, potency, and purity. Many CoAs report units like mg/g, %, ppm, or CFU. If a value falls outside the stated range, the CoA should include an explanation.
-
Look for sign-off: A dated signature from an authorized analyst or reviewer confirms the data was checked and approved.
If you’re ever unsure how to interpret a result, reach out to the brand’s support team for clarification. Clear documentation should make it easy to understand what the numbers mean.
How InfiniWell uses CoAs
At InfiniWell, Certificates of Analysis support a full-circle approach to quality. Every batch is tested, raw materials and finished products included, so you can compare real data with what you see on the label.
Here’s what that looks like:
-
Verified sourcing: We review CoAs for incoming raw materials before manufacturing begins. This includes identity, purity, allergen review, and other checks that support ingredient quality from day one.
-
Batch testing: Every production lot undergoes comprehensive testing at ISO/IEC 17025–accredited laboratories. Our current testing partners include MS Bioanalytical Labs and Eurofins, both known for validated, industry-standard methods.
-
Testing categories: Each batch is evaluated for identity, purity, potency, heavy metals, microbiological markers, allergens, and GMO status. These categories appear in our CoAs so you can see the exact results that support product quality and safety.
-
Stability and finished-good testing: Beyond required checks, we also perform stability testing. This ensures products maintain their expected quality throughout their shelf life, a 360-degree approach to testing that covers both raw materials and finished goods.
-
Documentation and access: We make CoAs publicly available. You can access them directly on each product page or request one from our support team if you need a specific batch.
What testing categories are included in an InfiniWell CoA?
An InfiniWell CoA reflects the full set of tests performed at accredited laboratories, which typically include:
-
Identity: Confirms the ingredient is exactly what the label states
-
Purity: Checks for unwanted substances or impurities
-
Potency: Verifies the amount of each active ingredient matches the specification
-
Heavy metals: Screens for elements like lead, arsenic, cadmium, and mercury
-
Microbiological markers: Ensures levels of microbes fall within acceptable limits
-
Allergens: Confirms the presence or absence of common allergenic compounds
-
GMO status: Indicates whether ingredients contain genetically modified material
These categories give a complete picture of product quality and help you evaluate the numbers quickly and confidently.
Turn CoA knowledge into smarter product choices
A CoA turns marketing claims into measurable facts. Once you know how to read one, you can compare formulas based on real data and choose products that align with your needs.
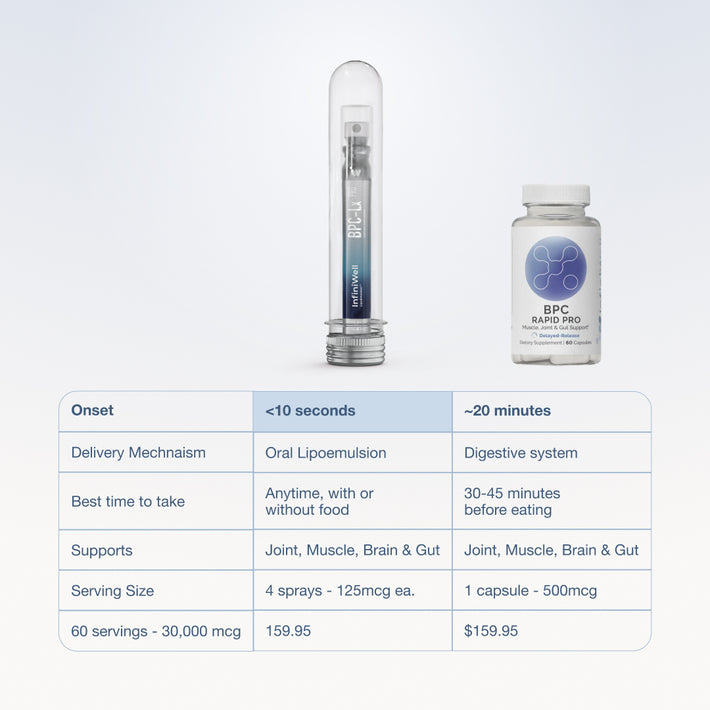
If you want an example of clear, batch-specific testing, start with BPC-Lx Pro, which features third-party verification from ISO-accredited facilities. Identity, potency, purity, microbiological markers, allergens, and heavy metals are all reviewed so you can see exactly how the product performs.
Looking for additional options? InfiniWell offers a full lineup of carefully formulated offerings, each supported by strong quality and testing practices.
For batch-specific information, visit each product page or request a CoA directly from the InfiniWell support team.
This content is for general informational purposes only and is not medical advice. Always talk to a qualified healthcare professional before starting any new supplement or making changes to your wellness routine.
What is a CoA: FAQs
Is a CoA the same as third-party testing?
Not exactly. Third-party testing refers to the process: an independent laboratory performs the analysis so the results aren’t influenced by the company that made the product. A CoA is the document that reports those results.
A CoA can come from either an in-house lab or a third-party lab, but results from an independent, ISO-accredited laboratory are generally considered more reliable because they’re unbiased and follow validated methods. A well-designed CoA also makes it clear which lab performed the testing, which helps you verify the source of the data.
The best case is when a brand uses a qualified third-party lab and provides a clear CoA so you can review the numbers yourself.
Where can I find CoAs for InfiniWell products?
InfiniWell posts CoAs on the individual product pages whenever they’re available. If you need a CoA for a specific batch, you can request it directly from our support team. We test every batch, so a matching document will always be on file.
All testing is performed at ISO/IEC 17025–accredited laboratories, including MS Bioanalytical Labs and Eurofins. These labs evaluate each batch for identity, purity, potency, heavy metals, microbiological markers, allergens, and GMO status. Those categories appear in the CoA so you can review the full set of results.
If you have questions about finding a CoA or interpreting the results, our support team is always available to help.
What's the difference between a CoA and an SDS?
A safety data sheet focuses on safe handling, storage, and transport, essentially worker safety and logistics. A CoA reports the test results for a specific batch and shows whether the product met its specification.
They’re complementary documents and often move through the manufacturing process together, but they serve different purposes.
Does every supplement need a CoA?
Practices vary, but a CoA is widely expected for raw materials and is increasingly common for finished products. Seeing a current, batch-specific CoA is a strong signal of quality control and good manufacturing practice culture.
-
SOURCES